White Papers
Synthetic Polyisoprene (IR) For Medical Products
September 20, 2020
Synthetic polyisoprene has become an important polymer for medical fluid control components Natural Polyisoprene (NR) and Synthetic Polyisoprene (IR) are rubber thermoset materials, which have properties that are very useful in many applications, especially medical fluid control components and device assemblies. These material properties include extremely high elongation, high values of tensile and tear resistance while also exhibiting excellent resilience, or the ability to quickly return or recover to the original shape after deformation.
Such properties make the use of Synthetic Polyisoprene (IR) ideal in medical applications requiring the prevention of fluid leakage around surgical instruments, fluid control valves re-sealing against a seat or interface, and pressure regulating membranes;

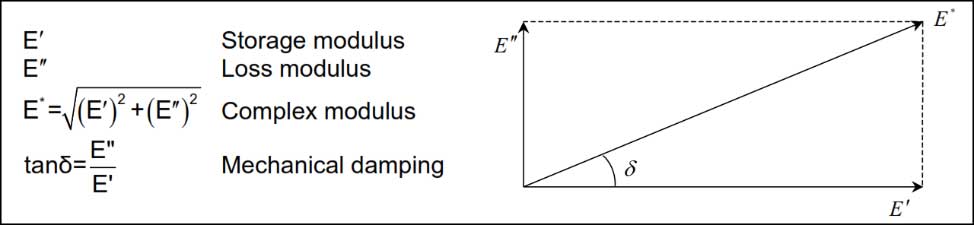
One may notice that all of these applications combine dynamic and static sealing requirements. In addition to all the typical material properties, Vernay Scientists realized they needed to look deeper. Resilience is a good example, which is actually a dynamic material characteristic. In evaluating and measuring the dynamic characteristics of materials, we frequently use a Dynamic Mechanical Analyzer (DMA) to quantify dynamic properties and compare different rubber recipes in effof rubber thermoset materials orts to optimize characteristics of resilience. Using the DMA, we can quantify the dynamic complex modulus, the storage modulus and the loss modulus. Through years of research, we have observed that maximizing the storage modulus and minimizing the loss modulus results in a more resilient recipe with reduced hysteresis. Think of the storage modulus as the spring component of the complex modulus, in some applications, it is preferred that the rubber part exhibits characteristics like an ideal spring – quick to respond and returning nearly all the energy (not absorbing or dampening energy). The characteristics of the rubber recipe are adjusted using various polymers, fillers, plasticizers and other ingredients, specifically to adjust the ratio of elastic vs loss modulae. This allows Vernay Chemists to match the resulting rubber material properties specifically to the application and part criteria.
In addition to the physical characteristics of the IR formulation, meeting specific industry regulations for medical applications is critical. These regulatory aspects include, but are not limited to; biocompatibility (USP Class VI and ISO10993), low extractable pharmaceutical and in some cases white listed materials (FDA 21CFR177.2600). Within these regulations are strict limitations on performance characteristics and laboratory tests that the materials must meet in areas such as; cytotoxicity, hemolysis, mutation, sensitization, limitations on maximum ingredient ratios, restricted ingredients, etc. Translating these requirements into the science and chemistry requires further consideration of impacts to material viscosity, rheology and resulting physical properties like modulus, resilience and chemical compatibility. It’s a delicate balance.
Material formulation and properties are important but they are not the only factor. The elastomeric materials and molded parts are dependent upon a balance of 3 critical technical aspects; 1) formulamedical tion material properties, 2) part/tooling geometry and 3) the molding process. We refer to these as the development “trifecta” because each one is dependent upon the others. Changes to any one will have impacts on the remaining two. Although Synthetic Polyisoprene (IR) exhibits many beneficial material properties and characteristics, the part geometry and molding process needs to be considered and balanced together in order to produce a successful product meeting all of the application criteria.

Vernay Laboratories has been a technology leader in the design, development, and manufacture of customized flow-control components since its founding in 1946. The company also formulates its own compounds to address each specific application.
Vernay’s products play a vital role in millions of medical devices, automobiles, and consumer products. Today, the privately held firm produces more than 1 billion components per year, drawing from a library of more than 33,000 elastomeric materials and compound formulations. It operates six plants around the world, in the U.S., the Netherlands, Italy and China. For more information, visit www.vernay.com.